 Note that each block is able to hold two electrons.Īn orbital diagram helps to determine the electron configuration of an element. Orbital diagram: The positions of the first ten orbits of an atom on an energy diagram. Within each energy level, the s orbital is at a lower energy than the p orbitals. The first energy level contains only one s orbital, the second energy level contains one s orbital and three p orbitals, and the third energy level contains one s orbital, three p orbitals, and five d orbitals. There are several different orbital shapes-s, p, d, and f-but we will be focusing mainly on s and p orbitals for now. These paths are called atomic orbitals, or subshells. Though electrons can be represented simply as circling the nucleus in rings, in reality, electrons move along paths that are much more complicated. The first 2 electrons are found in the first energy level, and the other 7 are found in the second energy level. The first two electrons are found in the first energy level, and the third electron is found in the second energy level.Īs an example, fluorine (F), has an atomic number of 9, meaning that a neutral fluorine atom has 9 electrons. The energy levels are shown as concentric circles around the central nucleus, and the electrons are placed from the inside out. The arrangement of electrons in a lithium atom: Lithium (Li) has an atomic number of 3, meaning that in a neutral atom, the number of electrons will be 3. The second shell can accommodate 2 x (2) 2, or 8, electrons. For example, the first shell can accommodate 2 x (1) 2 or 2 electrons. An atom's electron shell can accommodate 2n 2 electrons, where n is the energy level. Electrons further away from the nucleus will have higher energy. Electrons that are in the first energy level (energy level 1) are closest to the nucleus and will have the lowest energy. 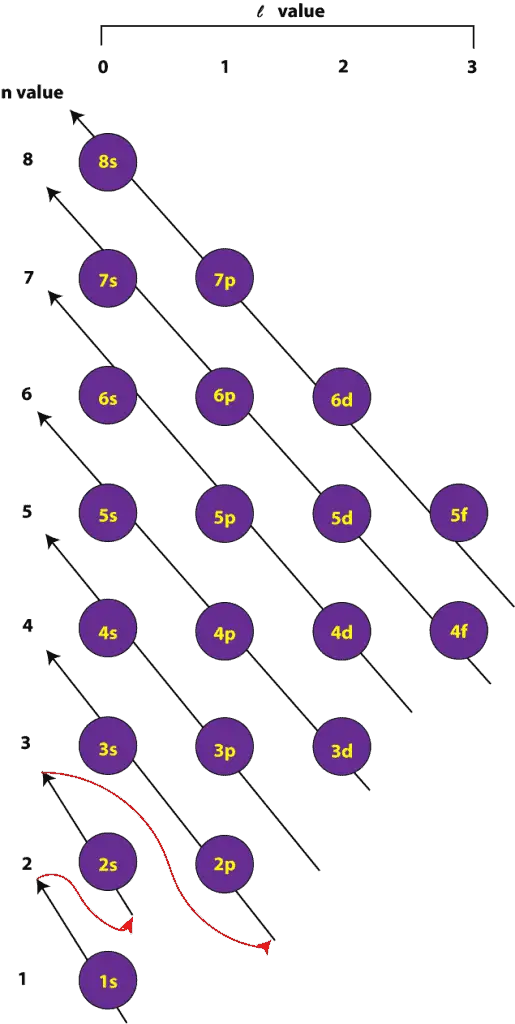
Here, electrons are arranged in energy levels, or shells, around the nucleus of an atom. We will start with a very simple way of showing the arrangement of electrons around an atom. The energy of the light released when an electron drops down from a higher energy level to a lower energy level is the same as the difference in energy between the two levels. This separating of electrons into energy units is called quantization of energy because there are only certain quantities of energy that an electron can have in an atom. The same goes for electrons and the amount of energy they can have. You need to lift your foot to the height of the step to move on. If you don't lift your foot enough, you will bump into the step and be stuck on the ground level. It is helpful to think of this like going up a flight of steps. Each element emits light at a specific frequency (or color) upon heating that corresponds to the energy of the electronic excitation. One way an electron can release energy is by emitting light. For the excited electron to go back to its original energy, or ground state, it needs to release energy. When the energy of an atom is increased (for example, when a substance is heated), the energy of the electrons inside the atom is also increased-that is to say, the electrons get excited. Electrons that have higher energy are found further away. Electrons with the lowest energy are found closest to the nucleus, where the attractive force of the positively charged nucleus is the greatest. Although these electrons all have the same charge and the same mass, each electron in an atom has a different amount of energy. The central structure of an atom is the nucleus, which contains protons and neutrons. quantization: The process of approximating a continuous signal by a set of discrete symbols or integer values.frequency: The number of occurrences of a repeating event per unit of time.Electrons will separate as much as possible within a shell. Each orbital can hold only one electron pair. An electron will move to the orbital with lowest energy. There are guidelines for determining the electron configuration of an atom.An orbital diagram is used to determine an atom's electron configuration. Within each shell, the s subshell is at a lower energy than the p. There are four different orbital shapes: s, p, d, and f. In a more realistic model, electrons move in atomic orbitals, or subshells.An atom's electron shell can accommodate 2n 2 electrons (where n is the shell level). 
Electrons closest to the nucleus will have the lowest energy. Viewed simply, electrons are arranged in shells around an atom's nucleus. 
The energy of the light released when an electron drops in energy level is the same as the difference in energy between the two levels. To go back to its ground state, the electron releases energy. If the energy of an atom is increased, an electron in the atom gets excited. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
